Kinetics is the study of the rate of chemical processes. There are 3 different orders of rates: zeroth, first, and second. Second rate reactions can be once again split into those with a singular reactant and those with two reactants.
The functional rate laws for these orders are found by integrating the differential rate laws based on the disappearance of reactant A and the concentration of product B. This is called the integrated rate law.
The integrated rate law for zeroth, first, and second order reactions are the following :
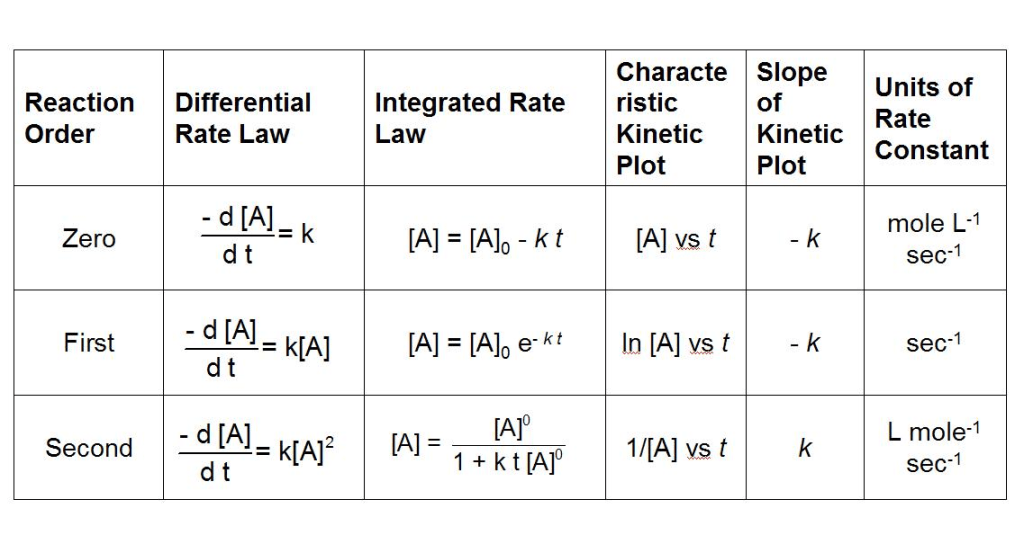
The rate constant k is one of few chemical constants that have varying units in different cases. These rate laws can be used to find the half life of various reactants in a reaction, calculated theoretical product yield at a certain time point, and even determine the rate determining step of a process with various intermediates.